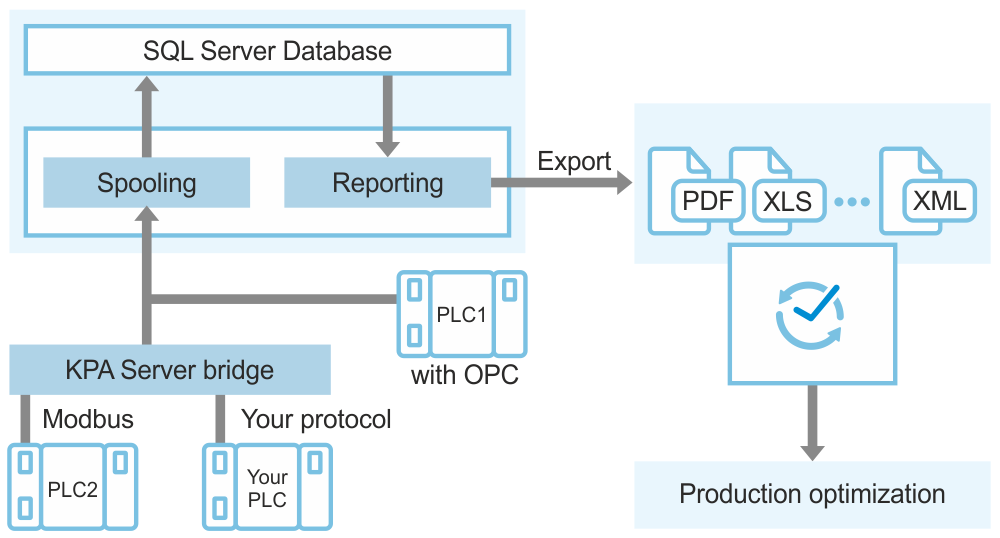
KPA Automation Data Analytics is an innovative reporting solution. It is suitable for companies of any size that need to collect a range of tag values in a table report for further analysis to control stability and optimize production.
Report customization
Reports are generated using selected tags and specially designed report templates, which can be customized according to individual company needs.
New Features in versions
Unlimited number of reports
KPA Automation Data Analytics has no limits on the number of created reports. A period for data selection can be set for each report.
Various report formats
Generated reports can be exported in HTML, PDF, and CSV file formats.
Scheduled report generation
KPA Automation Data Analytics allows to set specific time for automatic report generation.
User access control and logging
KPA Automation Data Analytics provides a wide range of user access settings. All user actions are logged.
Connection to various vendors PLC
Data is transferred between PLC and KPA Automation Data Analytics through the OPC UA protocol. Other protocols are supported using KPA Automation Server.
Suitable for CFR 21 Part 11 compliant systems
Title 21 Part 11 of the Code of Federal Regulations (CFR) establishes the United States Food and Drug Administration (FDA) regulations on electronic records and electronic signatures. CFR 21 Part 11 applies to medical device manufacturers, biotech companies, biologics developers, and other FDA-regulated industries.
Delivery package
KPA Automation Data Analytics is provided as a standalone application. Its delivery package includes the following software:
- KPA Automation View is used to select tags to be collected, configure reports, and view ready reports. There is no need to keep KPA Automation View running for data collection. All data is gathered by spooling service.
- Spooling and report generation services running in the background. Spooling service is used for sending OPC tag values to a database. Report generation service connects to the database and get these OPC tags values to create reports.
- KPA Automation Server (optional) is used for transferring data through protocols other than OPC UA.